Source: DEA
Review Of The Drug Enforcement Administration's (DEA)
Control Of The Diversion Of Controlled Pharmaceuticals
Report Number I-2002-010
September 2002
The Drug Abuse Prevention and Control Act (Act) was enacted on October 27, 1970. The Act initiated the "war on drugs" and focused on stemming the rising tide of illicit drug abuse and associated violence. Title II of the Act, known as the Controlled Substances Act (CSA), gave the Bureau of Narcotics and Dangerous Drugs (BNDD) the authority to regulate pharmaceuticals. The BNDD developed the policies and procedures for what was to become the OD when the DEA was established on July 1, 1973. Today, the OD regulates the distribution system for controlled pharmaceuticals and regulated chemicals, and is charged with preventing the diversion of those substances.
Over the past 30 years, controlled pharmaceutical use has dramatically increased. From 1973 to 2002, the number of controlled pharmaceuticals approved for sale by the Food and Drug Administration increased from 2,036 to 15,776. In 1970, individuals spent $5.5 billion on controlled pharmaceuticals in the United States. By 1999, this expenditure had increased to $99.6 billion.
With the rise in the manufacture and sale of controlled pharmaceuticals came the inevitable abuse and diversion of these beneficial drugs. According to the Acting Administrator of NIDA, the incidence of non-medical use of controlled pharmaceuticals has doubled over the last 10 years. The DEA reports that controlled pharmaceutical abuse now accounts for 30 percent of all reported deaths and injuries associated with drug abuse. According to the DEA, the most commonly diverted substances are narcotics, stimulants, depressants, and anabolic steroids. Additionally, HHS's Drug Abuse Warning Network (DAWN) study on overdose deaths shows controlled pharmaceuticals are factors in 25 percent of all reported overdose deaths and 20 percent of all emergency room visits relating to drug abuse. Further, as shown in Chart 1 on the next page, over the past 15 years the estimated number of first-time abusers of pain relievers, tranquilizers, stimulants, and sedatives has increased.
| Chart 1. Estimated Number (in Thousands) of First-Time Non-Medical Users of Certain Categories of Controlled Pharmaceuticals, 1985-1999
[Image is not available electronically.]
|
According to a September 2001 DEA report, hydrocodone products remain the most commonly diverted category of controlled pharmaceuticals.3 All 21 DEA domestic field division offices mentioned this drug in their third quarter fiscal year (FY) 2001 reports as one of the most commonly diverted controlled pharmaceuticals. Hydrocodone diversion has been escalating over the past decade. In 1994, 7 million dosage units were illegally diverted; this increased to 11 million in 1997. Since 1990, the number of hydrocodone prescriptions increased by 300 percent, while during the same period emergency room visits attributed to hydrocodone abuse increased by 500 percent. OxyContin diversion has also become an increasing problem.4
Diversion Control Program Organization and Staffing
The OD is located at DEA Headquarters in Washington, D.C. It provides policy direction, program guidance, and support to DEA diversion staff in the field. The OD is a subcomponent of DEA's Operations Division, which is one of six major organizational elements at DEA Headquarters. DEA special agents head both the Operations Division and the OD. According to the OD Deputy Assistant Administrator, a proposal to elevate the OD to a division is currently under review by DEA's Office of Legal Counsel and the Department of Justice (DOJ).
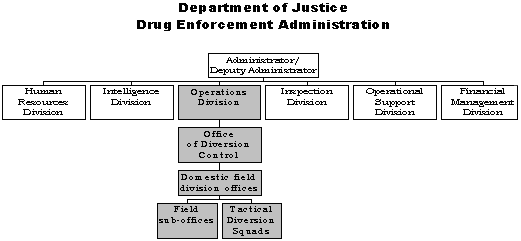
The DEA field structure includes 21 domestic field division offices headed by a Special-Agent-in-Charge (SAC) and 214 sub-offices. Although the staff at DEA field offices are predominantly DEA special agents, with full law enforcement authority, every field division office and 51 of the sub-offices have a diversion control unit. These units are staffed by diversion investigators, who do not have law enforcement authority, and are typically headed by a diversion investigator supervisor. Diversion program managers at each of the 21 field divisions supervise diversion control operations within their geographical area of responsibility.
Figure 1. Organizational Chart for Drug Enforcement Administration

Source: DEA
The DEA also has established Tactical Diversion Squads (TDS) at selected field locations. As of July 2002, TDSs were located at field offices in Boston, Denver, Houston, New Orleans, Seattle, and St. Louis. The TDSs consist of a combination of federal, state, and local law enforcement officers. The mission of these multi-agency squads is to detect, investigate, disrupt, and refer for prosecution violators of the CSA and similar state statutes; in short, to curtail the diversion of controlled pharmaceuticals and regulated chemicals within a geographic area. The TDSs currently operate under the supervision of a special agent, but were formerly supervised by a diversion program manager.
Diversion investigators represented 10 percent, or 523, of the DEA's 5,124 authorized investigator positions in FY 2001. The authorized diversion investigator positions were assigned as follows: 55 at headquarters, 455 at domestic field offices, and the remaining 13 at overseas offices. At the end of FY 2001, actual on-board staffing consisted of 43 diversion investigators at OD headquarters and 424 diversion investigators at DEA domestic field and overseas offices.
Diversion Investigator Responsibilities
The CSA requires all businesses that manufacture or distribute controlled pharmaceuticals; all health professionals who dispense, administer, or prescribe controlled pharmaceuticals; and all pharmacies that fill prescriptions to register with the DEA. At DEA field offices, diversion investigators are responsible for investigating applications of potential registrants, monitoring existing registrants through cyclical investigations, and investigating allegations of the diversion of controlled pharmaceuticals and regulated chemicals. According to the DEA's Work Hours Reporting System, during FY 2001 field diversion investigators spent 20 percent of their time on processing registrant applications, 13 percent on cyclical investigations, 66 percent on investigations of criminal complaints or alleged criminal activity, and 1 percent on other activities.5
Diversion investigators in DEA field offices review and approve applications submitted by potential registrants. Prior to approving an application, diversion investigators conduct a background review of the criminal histories of the applicant, the applicant's company, and company employees; ensure the applicant has not had similar federal or state licenses revoked in the past; and inspect the applicant's security measures to protect the controlled pharmaceuticals from theft. Once approved, the applicant is assigned a DEA registration number and is permitted to manufacture and/or distribute controlled pharmaceuticals.
As part of the registrant monitoring process, diversion investigators conduct investigations of registrants every five years to ensure they are complying with federal law and regulations. For example, during a review of a drug manufacturer the diversion investigator ensures that the amounts of specific controlled pharmaceuticals produced are within the DEA's prescribed limits and that the manufacturer is complying with regulatory requirements relating to physical security, records accountability, and adherence to CSA standards. Violations of regulations may result in administrative, civil, or criminal action, depending on the severity of the infraction.
With respect to investigations of diversion of controlled pharmaceuticals, examples of DEA cases include:
Diversion investigators learn of possible diversion of controlled pharmaceuticals through information received from the public, local police, or DEA informants. Because diversion investigators do not have arrest authority or carry weapons, they must rely on the assistance of DEA special agents or other law enforcement officers, such as state or local police, to assist them in their investigations.
Diversion Control Program Funding
Public Law 102-395 required that effective October 1, 1993, the DEA collect fees (both initial and renewal fees) to ensure the recovery of the full costs of operating the OD. The legislation required that registration fees collected by the DEA be deposited into a Diversion Control Fee Account within the general fund of the U.S. Treasury. At least quarterly, the U.S. Treasury is required to provide funds from this account to reimburse the DEA for expenses involving controlled pharmaceutical diversion operations.
The registrant fees collected by the DEA support only the registering, monitoring, and investigating activities associated with controlled pharmaceuticals. The other function of the OD - registering, monitoring, and investigating activities associated with regulated chemicals - is funded by direct appropriations. In FY 2001, the DEA expended approximately $65.7 million out of its registrant fee account for controlled pharmaceutical diversion operations. During this same period, the DEA was appropriated $16.1 million to fund its regulated chemical diversion operations.
Serious problems with OxyContin abuse were initially noticed in 1998 in depressed rural eastern areas of the United States including sections of Maine, Virginia, and Kentucky. By the early fall of 2000, OxyContin abuse rapidly increased and spread to other areas of the country, especially in Maryland, West Virginia, Florida and urban areas such as Philadelphia, Pennsylvania and Boston, Massachusetts. Among other places, OxyContin has been identified as a serious problem in Arizona, Louisiana, and Ohio.